Valence Number is the number of valence electrons in an atom or combined group of atoms which can be easily given up or accepted to react with or bond to another atom or group of atoms to form a molecule. It is a whole number (positive or negative) which represents the power of one element to combine with another. Administrative Offices: 1900 West Polk Street, Suite 220C. Chicago, IL 60612. In chemistry, the valence or valency of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
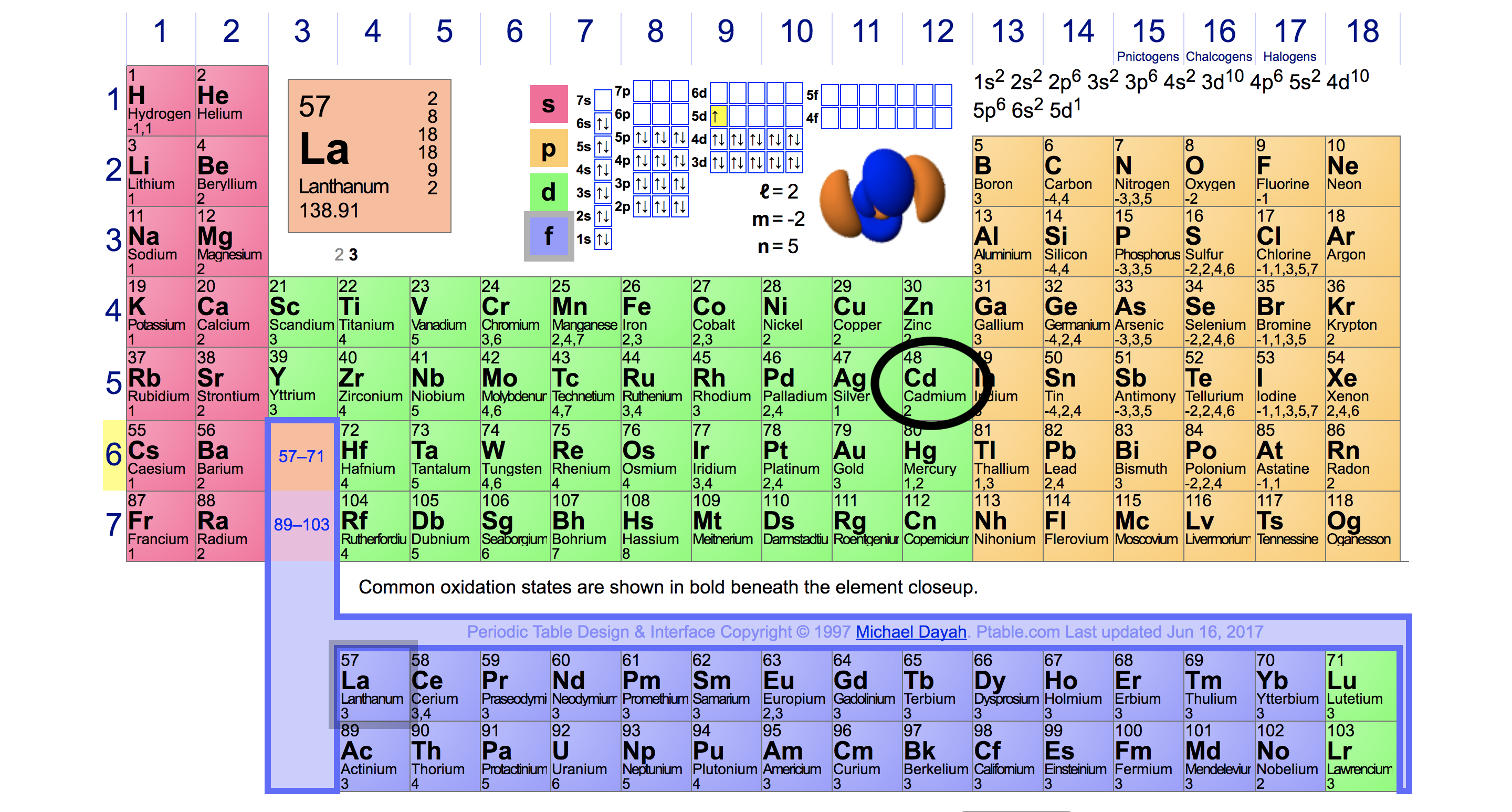
However, they do not duplicate the results that we derived from the method above. One cannot enumerate their valence electrons by simply referring to their group number. This boils down to the way electrons occupy shells in transition elements.
The solar system analogy describing the arrangement of electrons around an atom is completely false. It should be immediately eliminated, but because it alleviates the difficulty that accompanies the exposition of the Electrons do not occupy rigid shells around their nucleus. In fact, their location around a nucleus is highly uncertain. They can only occupy distinct energy levels around a nucleus. They’re There’s a rule that restricts the number of electrons that a sub-shell can accommodate: s-2, p-6, d-10 and f-14. If this wasn’t enough, adding to the delirium, the shells can only be filled in a specific order given below. Let’s call it The electrons must be filled from left to right in this exact order only. However, because we must abide by 4s must be filled before 3d, such that there are now 8 in the 3th making the configuration: 2,8,8,2. Voila! As Richard Feynman would cheerfully exclaim: The pleasure of finding things out! Sadly, the joy is only half-lived — the reason for Okay, so now that we know how shells are filled, we can move further to find the number of valence electrons in the transition elements. Consider Scandium (Sc) with its atomic number of 21. Filling the electrons according to our st electron occupies the 3d sub-shell. However, as the previously filled 4Similarly, every transition element in the 4th shell are the inhabitants of the outermost shell and rightfully deserve the designation of valence electrons. th
Valence Number Carbon

, in conclusion, the number of valence electrons for transition and inner transition elements varies in an unpredictable manner.
The capricious behavior of their valence electrons, interminably quivering and hopping in indecision, deny any attempt to obtain a singular stable configuration — predicting then the number of valence electrons is near to impossible!
Boron Valence Number